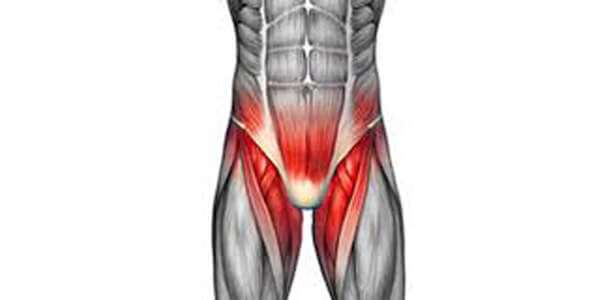
Topol GA, Reeves KD: Regenerative injection of elite athletes with career-altering chronic groin pain who fail conservative treatment: a consecutive case series. Am J Phys Med Rehabil 2008;87; – .
Dr. Reeves’ Notes: Dextrose 12.5% injection returns 66/72 elite rugby and soccer players to full sport who were prevented from high level play by chronic groin pain. This was a continuation of the consecutive patient study by Topol et al in 2005 with durability of benefit confirmed by up to 6 year followup.
An abstract of this study on Regenerative Injection of Elite Athletes is available here, with a copy of the content below.
OBJECTIVE:
To obtain multisport and long-term outcome data from the use of regenerative injection therapy on career-threatened athletes.
DESIGN:
Consecutive enrollment of elite performance-limited athletes with chronic groin/abdominal pain who failed a conservative treatment trial. The treatment consisted of monthly injections of 12.5% dextrose in 0.5% lidocaine in abdominal and adductor attachments on the pubis. Injection of the nociceptive source was confirmed by repetition of resistive testing 5 mins after injection.
RESULTS:
Seventy-five athletes were enrolled. Seventy-two athletes (39 rugby, 29 soccer, and 4 other) completed the minimum two-treatment protocol. Their data revealed a mean groin pain history of 11 (3–60) mos. Average number of treatments received was 3 (1–6). Individual paired t tests for Visual Analog Scale (VAS) of pain with sport (VAS Pain) and Nirschl pain phase scale measured at 0 and an average of 26 (6–73) mos indicated VAS Pain improvement of 82% (P <10 10 ). and Nirschl pain phase scale improvement of 78% (P <10 10 ). Six athletes did not improve following regenerative injection therapy treatment, and the remaining 66 returned to unrestricted sport. Return to unrestricted sport occurred in an average of 3 (1–5) mos.
CONCLUSIONS:
Athletes returned to full elite-level performance in a timely and sustainable manner after regenerative injection therapy using dextrose.
Topol GA, Reeves KD, Hassanein K. Efficacy of Dextrose Prolotherapy in Elite Male Kicking-Sport Athletes With Chronic Groin Pain. Archives Phys Med Rehabil, 2005;86:697-702.
Dr. Reeves’ Notes: Dextrose 12.5% injection returns 22/24 elite rugby and soccer players to full sport who were prevented from high level play by chronic groin pain. This involved treatment of abdominal muscle weakness, adductor sprain and osteitis pubis.
An abstract of the study on Prolotherapy in Elite Male Kicking-Sport Athletes is available here, with a copy of the content below.
PARTICIPANTS:
Twenty-two rugby and 2 soccer players with chronic groin pain that prevented full sports participation and who were non-responsive both to therapy and to a graded reintroduction into sports activity.
INTERVENTION:
Monthly injection of 12.5% dextrose and 0.5% lidocaine in thigh adductor origins, suprapubic abdominal insertions, and symphysis pubis, depending on palpation tenderness. Injections were given until complete resolution of pain or lack of improvement for 2 consecutive treatments.
MAIN OUTCOME MEASURES:
Visual analogue scale (VAS) for pain with sports and the Nirschl Pain Phase Scale (NPPS), a measure of functional impairment from pain.
RESULTS:
The final data collection point was 6-32 months after treatment (mean, 17mo). A mean of 2.8 treatments were given. The mean reduction in pain during sports , as measured by the VAS, improved from 6.3 +/- 1.4 to 1.0 +/- 2.4 (P < .001), and the mean reduction NPPS score improved from 5.3 +/- 0.7 to 0.8 +/- 1.9 (P < .001). Twenty of 24 patients had no pain and 22 of 24 were unrestricted with sports at final data collection.
CONCLUSIONS:
Dextrose prolotherapy showed marked efficacy for chronic groin pain in this group of elite rugby and soccer athletes.
AUTHORS:
Gastón Andrés Topol, M.D. Physical Medicine and Rehabilitation Service Jaime Slullitel Rosario Orthopedic and Trauma Institute, Argentina. Team Physiatrist, Rosario Rugby Union, K. Dean Reeves, M.D., FAAPM&R Associate Clinical Professor, Dept of PM&R, Univ. of Kansas Medical Center, and Khatab Mohammed Hassanein, Ph.D. Professor and Chairman Department of Biometry University of Kansas Medical Center, Kansas City, Kansas.